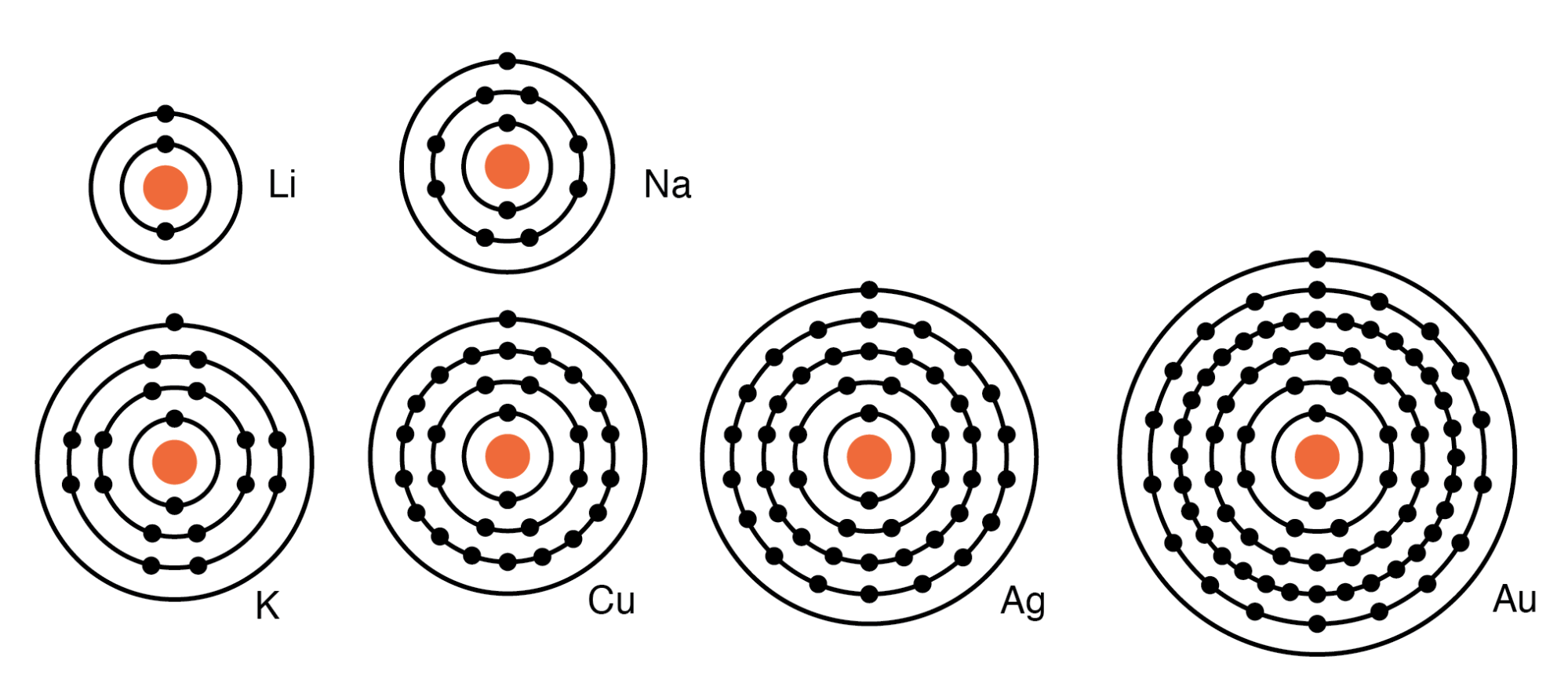 
The electron configuration shows that sodium ion(Na +) have only two shells and the last shell has eight electrons. Na e Na + The electron configuration of sodium ion(Na +) is 1s 2 2s 2 2p 6. The orbitals are filled as described by Hund’s rule: the lowest-energy configuration for an atom with electrons within a set of degenerate orbitals is that having the maximum number of unpaired electrons. Sodium donates the electron of the last shell to form bonds and turns into a sodium ion(Na +). We now have a choice of filling one of the 2 p orbitals and pairing the electrons or of leaving the electrons unpaired in two different, but degenerate, p orbitals. This means that B has two valence electrons in 3s (3s 2. Element B is located in Period 3, the 2nd position in 3s-block. Two electrons are needed (Na and Mg) to fill the 3s subshell and six electrons are required (Al through Ar) to complete the 3p. Sodium cation loses one electron, so Na +: 1s 2 2s 2 2p 6 3s 1. For transition metals, the last s orbital loses an electron before the d orbitals. List of unique identifiers for Sodium in various chemical registry databases. For main group elements, the last orbital gains or loses the electron. The remaining two electrons occupy the 2 p subshell. The third row is similar to the second row elements. Remember electrons are negatively charged, so ions with a positive charge have lost an electron. Four of them fill the 1 s and 2 s orbitals. When drawing orbital diagrams, we include empty boxes to depict any empty orbitals in the same subshell that we are filling.Ĭarbon (atomic number 6) has six electrons. 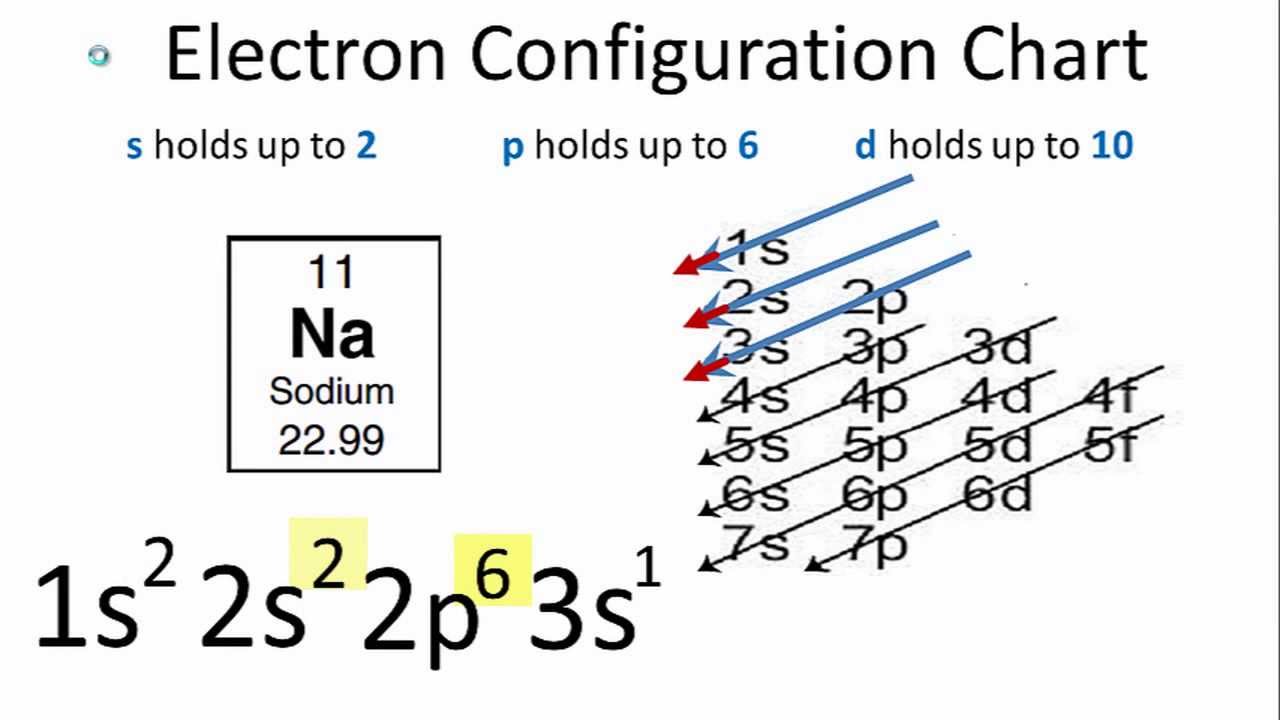
There are three degenerate 2 p orbitals ( m l = −1, 0, +1) and the electron can occupy any one of these p orbitals. Because any s subshell can contain only two electrons, the fifth electron must occupy the next energy level, which will be a 2 p orbital. The n = 1 shell is filled with two electrons and three electrons will occupy the n = 2 shell. The fourth electron fills the remaining space in the 2 s orbital.Īn atom of boron (atomic number 5) contains five electrons. A beryllium atom, with two valence electrons, has the electron dot diagram below: Since electrons repel each other, the dots for a given atom are distributed evenly around the symbol before they are paired. To find the number of neutrons, you will need to subtract the atomic number from the atomic mass. Subtract the atomic number from the atomic mass. Step 2: Identify the electron of interest, and ignore all electrons in higher groups (to the right in the list from Step 1). For example, the atomic mass of boron is 10.811, but you can just round the atomic mass up to 11. Step 1: Write the electron configuration of the atom in the following form: (1s) (2s, 2p) (3s, 3p) (3d) (4s, 4p) (4d) (4f) (5s, 5p). Thus, the electron configuration and orbital diagram of lithium are:Īn atom of the alkaline earth metal beryllium, with an atomic number of 4, contains four protons in the nucleus and four electrons surrounding the nucleus. Electron dot diagrams are diagrams in which the valence electrons of an atom are shown as dots distributed around the elements symbol. Make sure that you round the atomic mass to the nearest whole number. An interactive periodic table may be found here.\) ). If an electron was the mass of a penny, a proton or a neutron would have the mass of a large bowling. 
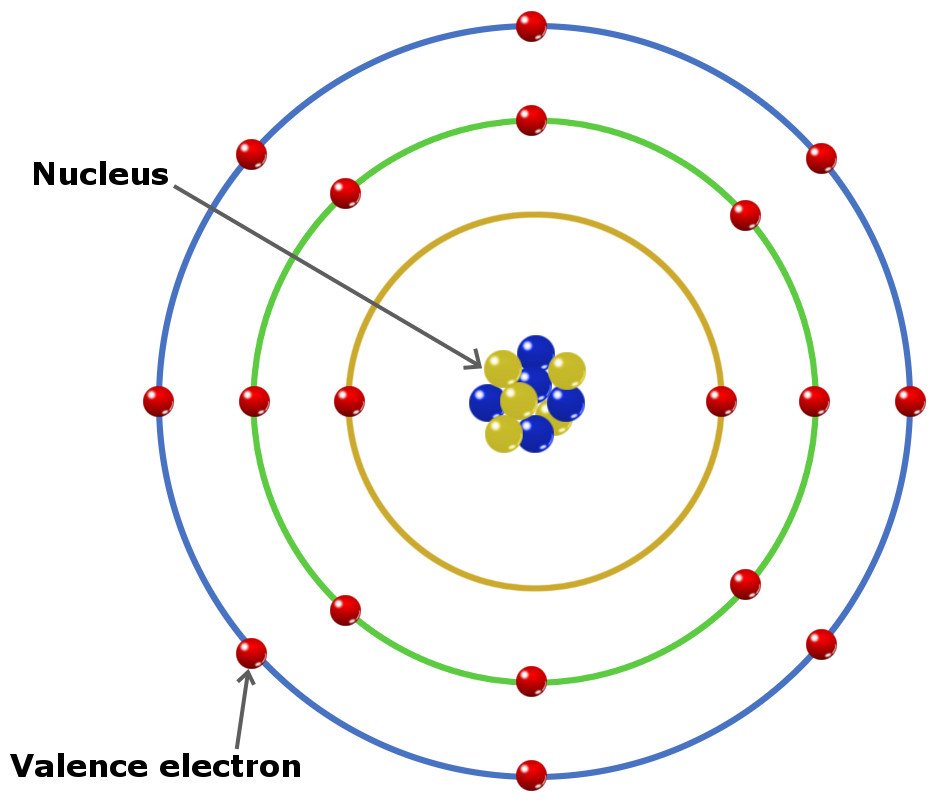
1: Electrons are much smaller than protons or neutrons. \): The modern version of the periodic table. While protons and neutrons are located inside the nucleus at the center of the atom, electrons are located outside the nucleus in what is often called the electron cloud.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
